

Surgical treatments for benign enlargement of the prostate (BPH) are an option for men with troublesome symptoms who have not had satisfactory results after trying prostate medication, or who do not wish to remain on longterm medication for their prostate condition. Surgery is also used to treat men suffering from urinary retention who are unable to pass urine and are dependent on a catheter to empty their bladder, and for men who have repeated urine infections or bleeding due to their enlarged prostate.
REZUM is the new minimally invasive treatment that uses steam vapour to shrink the prostate and improve urinary symptoms. It is carried as day case and patients are able to return normal daily activity within a few days.
Uro Lift is a new minimally invasive treatment for men with urinary symptoms due to minimal- moderate enlargement of the prostate. It is performed from within the urethra and involves pinning back rather than removing the obstructive prostate tissue. It is not suitable for all men with urinary symptoms and is as yet unproven in the treatment of men with urinary retention. Advice from a specialist urologist, such as those in the Cambridge Urology Partnership, is advised in order to determine whether UroLift might be suitable for you.
HoLEP (Holmium laser enucleation of the prostate) is a minimally invasive surgical treatment for men with any degree of prostate enlargement who have urinary symptoms, and men who are suffering from retention of urine, repeated urine infections or bleeding due to their enlarged prostate. Like urolift, it is performed from within the urethra. A laser is used to remove the area of the prostate that is causing the problem.
TURP (Transurethral resection of the prostate) is also an effective procedure to treat men with benign enlargement of the prostate and this is also offered by Cambridge Urology Partnership. TURP is the traditional treatment which involves scraping away the part of the prostate tissue which is causing a blockage using an electric current.
By offering HoLEP, REZUM, UroLift and TURP, Cambridge Urology Partnership is able to offer patients a range of treatment options for their prostate problem, and you can be sure that you will receive the treatment which is most suited to you.
HoLEP, or Holmium Laser Enucleation of the Prostate was developed in New Zealand in the late 1990’s. It is normally done under general anaesthetic. The prostate is approached internally (endoscopically), through the urethra, without any cuts on the skin. The Holmium laser is used to peel out the obstructive core of the prostate in its entirety. This leads to maximal relief of prostate obstruction and a man will usually only require 1 HoLEP procedure in his lifetime. It is very unlikely that the prostate will regrow after HoLEP (0.7% need a re-operation up to 10yr after HoLEP compared to 10-15% after TURP, and more after green light laser surgery).
Diagram showing the complete removal of the obstructive inner part of the prostate that is achieved with HoLEP
After HoLEP, the area of the prostate that is removed is sent for microscopic analysis. Cancer that would otherwise be undetected is found in 8%.
HoLEP is one of the most extensively researched procedures for BPH. 16 HoLEP randomised trials (the highest quality of clinical trial) have been published up to Dec 2014. The clinical trials comparing it to the previous “gold standard” of TURP, consistently show advantages for HoLEP that include: Less bleeding, fewer blood transfusions, shorter time with a catheter in place after surgery, and shorter hospital stay (90% of men having HoLEP are able to leave the hospital without a catheter the day after surgery, and in some cases there is no need to stay in hospital overnight). Meta-analyses which are studies analysing the results of large numbers of randomised trials, suggest that HoLEP relieves urinary symptoms and improves urinary flow rates more effectively than all other endoscopic procedures for BPH. The average symptom score improvement after HoLEP is 70-80% and flow rate improvements of up to 600% have been reported. Recent studies have shown that HoLEP is extremely effective at treating men in urinary retention. 98% of men with catheters before surgery are freed from their catheter dependence after HoLEP. This compares to 70% with green light laser and TURP surgery. Unlike all other endoscopic procedures for BPH, HoLEP is suitable for men with any size prostate.
At Cambridge Urology Partnership we have four surgeons who perform HoLEP: Mr Tev Aho, Mr James Armitage, and Mr Christof Kastner, and Mr Kasra Saeb-Parsy between them they have one of the largest experiences in this technique in the UK, and are amongst the leading in the world. They regularly perform this surgery, as well as teach this technique to other surgeons, and lecture on its use in national and international meetings.
Click here to watch our video on HoLEP technique part one
Click here to watch our video on HoLEP technique part two
Click here to watch our video on HoLEP technique part three
Click here to watch our video on what to expect three months after HoLEP surgery
Click here to see Tev Aho explain about prostate enlargement and HoLEP surgery
What is Rezūm?
Rezūm is a method by which steam energy is used to ablate (or remove) the particular part of the prostate that enlarges and causes symptoms due to BPH. Specific technical information is explained below.
This system consists of a portable radiofrequency (RF) generator and delivery device that is introduced into the body via the urethra (transurethral approach) and guided by direct visualization through a telescopic lens placed within the delivery device.
RF energy from the generator is applied to an inductive coil in the delivery device to heat up a controlled amount of water outside of the body, converting the water into vapour or steam. The thermal energy created outside the body is delivered into the prostate tissue through a tiny needle with emitter holes to ablate the targeted obstructive prostate tissue that causes BPH. The procedure takes approximately 3 – 7 minutes and can be performed in a Day Surgery setting. No RF energy is delivered into the body.
Throughout the insertion of the device and during the treatment saline (salty water) is running to help ensure better views and to prevent the urethra from overheating.
The generator has a number of safety features to ensure proper heating and thermal ablation of the targeted prostate tissue, while protecting the urethra during treatment. Thermocouples in the delivery device monitor temperatures to ensure consistent delivery of thermal energy into the ablation region. The length of the vapour needle is fixed and the operator controls all functions of the device. Placement of the vapour needle within the prostate is done under direct visualization through the telescopic lens, which is in the urethra inside the prostate.
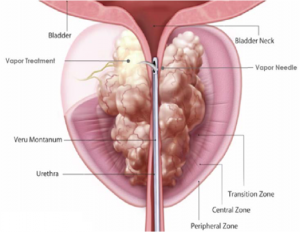
What approvals does Rezūm have?
The Rezūm System received CE Mark on 8th July 2013 and FDA clearance for use in the USA on 27th August 2015.
Rezūm received NICE approval in August 2018 and it has advised that the procedure may also be effective for patients with an enlarged median prostatic lobe.
What evidence of the benefits of Rezūm are available?
There have been several clinical studies undertaken on the benefits of the Rezūm System and we can give you details if you would like to review them.
Rezūm provides the following benefits:
Potential alternative to BPH medications
Relieves symptoms safely and effectively1
Provides noticeable symptom improvement within two weeks 1, maximum benefit 1-3 months
Preserves sexual and urinary functions1
Allows patients to return to regular activities within a few days1
What happens next?
You will be offered a date for the procedure and also for a pre-operative assessment if a general anesthetic is going to be used. If you have not already done a flow test, this will be performed as well as completing the questionnaires. If you are on blood thinners, you may be asked to stop taking them for a period prior to your treatment.
Treatment day
You will have been given a time to attend the hospital. Even though the procedure only takes a few minutes, you will probably be at the hospital some time before and after the procedure, again depending on the anesthetic used. It is very likely that you will be discharged home the same day.
The treatment
About an hour before, you will be given some strong painkillers as well as some antibiotics which you will continue at home. You will then be accompanied to theatre where the procedure will take place. If you are having a general anesthetic you will be sent to sleep.
The Rezūm equipment is then passed through the urethra using plenty of anesthetic gel, which may momentarily sting. After a quick inspection of the bladder, the treatment will begin.
On completion a catheter will be left within your bladder for a few days prior to voiding as the prostate readjusts to the treatment and the swelling subsides.
When can I go home?
After your procedure, you will be reviewed by a member of the urology team who will discuss the operation, any specific requirements for you at home and a plan for your aftercare.
A date will be given to you as when to return for the catheter removal as well as the care required whilst at home.
It is normal to have some pain or discomfort after operations, and we advise you on painkillers to help keep it under control.
Return to work as you feel comfortable depending on your job. If in doubt please check with your surgeon and obtain a certificate, before you are discharged
When you attend for catheter removal, the tube will be removed and then when you are ready you will be asked to void into the flow machine and the bladder scanner will detect any remaining volume of urine. If the nurse is happy with the results you will be discharged.
If there are any problems you may require a further period of catheterisation or be taught intermittent self catheterisation.
Within a few days you will be able to resume normal activities and should notice improvements within 2 weeks, although it may be 3 months before the full benefit is evident. Blood may be visible in the urine and ejaculation fluid
When can I resume sexual activity?
We recommend you avoid ejaculation for 6-8 weeks following the procedure.
1McVary KT, Roehrborn CG. Three-year outcomes of the prospective, randomized controlled Rezūm system study: Convective radiofrequency thermal therapy for treatment of lower urinary tract symptoms due to benign prostatic hyperplasia. Urology. 111C (2018):1-9.
Click here to watch a video REZUM treatment
NICE approval for REZUM (Medtech innovation briefing [MIB158])
The UroLift treatment is a new treatment . Typically it is suitable for patients who are suffering from urinary symptoms caused by a prostate gland which is only mildly to moderately enlarged, and may also be suitable for younger patients who require some surgery to the prostate for whom the possibility of sexual side effects from medication or the conventional surgery offered for this condition are unacceptable.
This procedure involves passing a fine telescope-like instrument into the urethra, or waterpipe. This device enables the surgeon to deploy a small implant between the inner and outer surfaces of the prostate, rather like a small treasury tag. This implant retracts the enlarged prostate lobe without the need for any cutting or burning of tissue The number of implants required varies according to the size and shape of the prostate, and usually between 2 and 4 implants need to be deployed. After the implants have been placed, most patients will be able to pass urine without the need for a catheter.
The operation can be performed under local anaesthetic or sedation or under a general anaesthetic, which means you will be asleep for the entire procedure. Typically, patients will go home on the same day as the surgery. The procedure usually does not require placement of a catheter afterwards.
Diagram showing the prostate prior to treatment with the UroLift device, and subsequent insertion of the device and the final situation with the procedure complete.
The average time to return to work or daily activities is 5 days . Also of note is that there have not been reported incidences of dry orgasms or erection problems.
There have been several publications in all the leading Urological Journals as well as FDA approval in 2013 and NICE approval in January 2014. The published evidence showed that in men treated with Urolift, the flow rate improved by 50% (up to twice that of drugs), symptoms improved by 50% (up to 2-3 times that seen with drugs) and no sexual dysfunction. Currently follow-up data is published to two years. There was no incidence of sexual dysfunction in any of the studies. 80% of men in the studies who had not had treatment with the device elected to undergo Urolift.
Click here to read our information sheet on UroLift (link to UroLift information sheet).
NICE interventional procedures guidance [IPG475] On Urolift January 2014
British Journal of Urology 2014 Multicentre prospective crossover study of the ‘prostatic urethral lift’ for the treatment of lower urinary tract symptoms secondary to benign prostatic hyperplasia.
Journal of Sexual Medicine 2014 Treatment of LUTS secondary to BPH while preserving sexual function: randomized controlled study of prostatic urethral lift.
Canadian Journal of Urology 2014 Prospective multi-center study elucidating patient experience after prostatic urethral lift.
European Urology 2013 Minimally invasive prostatic urethral lift: surgical technique and multinational experience.
Journal of Uology 2013 The prostatic urethral lift for the treatment of lower urinary tract symptoms associated with prostate enlargement due to benign prostatic hyperplasia: the L.I.F.T. Study.